|
1 Unite de Recherche en Genetique Moleculaire et
en Hematologie, TNSERM U.91 CN RS U A 607, Hopital Henri Mondor
, 94010 Creteil, France.
Most acutely transforming leukemogenic or sarcomagenic retroviruses have transduced in their genomes altered cellular genes named oncogenes [1]. These viruses are usually defective in replication as a result of the deletion of structural genes, and they require a helper virus for propagation. Isolation of new acutely transforming retroviruses, comprehensive studies of their physiopathological processes, and molecular analysis of their genomes remain, therefore, powerful means for the identification of key genes involved in the regulation of cell growth, differentiation, or development. The purpose of this paper is to summarize the data that we have accumulated over the past few years on a recently isolated acute leukemogenic murine retrovirus named myeloproliferative leukemia virus (MPL V).
We isolated the MPL V in 1985 at the Curie Institute (Orsay, France) during a research program designed to evaluate the in vivo transforming properties of different Friend helper viruses (F MuL V). While several clonal F-MuL V isolates have the capacity to induce a rapid erythroblastosis in newborninoculated NIH Swiss or BALB/c mice [2], DBA/2 mice were found to be resistant to this early erythroleukemia [3]. Nevertheless, they developed various types of hematopoietic malignancies after a latent interval of712 months [4-6]. In general, these leukemias ( either myelogenous, lymphoid, or erythroid) were associated with a more or less severe anemia. Out of 238 DBA/2 mice inoculated at birth with F -MuL V clone 57 [7], one mouse developed, after 7 months of infection, an hepatosplenomegaly unusually accompanied with a polycythemia. Cellfree extract prepared from the original leukemic spleen or supernatant medium from an in vitro permanent cell line derived from the leukemic spleen cells caused an explosive leukemia upon inoculation into adult mice of most strains, including C 57 Bl strains. The disease was characterized by hepa tosplenomegaly, polycythemia, pronounced myelemia but no thymus or lymph node involvement, and death within 1- 3 months. Spleen and liver were extensively infiltrated with maturing precursor cells belonging to the granulocytic, erythroblastic and megakaryocytic lineages. Typically, the blood of severely diseased animals was also massively invaded by morphologically normal polymorphonuclears, erythroblasts, and platelets. Several hematopoietic lineages were obviously involved in this disease, hence our name for the virus isolate, "myeloproliferative leukemia virus" [8].
Virologic studies of MPL V by Penciolelli et al. [9] demonstrated
that this highly leukemogenic virus isolate contained two dissociable
retroviral genomes: one was the parental replication-competent FMuL
V 57, and the second was a new replication-defective component now
designated as MPL V. A comparison of viral RN A species expressed
in F -MuLV alone or F-MuLV + MPLV-producing cells by Northern blot
analysis showed that MPLV was 0.8 kb shorter than F-MuLV and that
a deletion had probably occurred in the MPL V env gene. This was
further confirmed by the establishment of the MPL V restriction
endonuclease map which was compared with that of F-MuLV [10]. From
their data, these investigators concluded that the MPLVdefective
genome
In an attempt to define the origin and nature of the genetic sequences
contained in the MPL V -rearranged env region, Souyri et al. [11]
derived cDNA probes which were nonhomologous to sequences contained
in F-MuLV. Two probes were found to be MPL V specific, in that they
hybridized to RNA of MPLV -containing nonproducer cells but did
not hybridize to RNA ofecotropic MuLVs norto RNA of amphotropic
or xenotropic murine viruses. This indicated that, in contrast to
Friend spleen focus-forming viruses (SFFV), MPL V did not result
from a recombination between F-MuLV and a portion of the env gene
of murine xenotropic virus [ 12, 13] . A full-length biologically
active MPL V provirus was molecularly cloned from a genomic library
of a nonproducer Mus dunni clone [11 ]. Sequence analysis revealed
that the MPL V env gene contains a large open reading frame which
could code for a polypeptide of 284 amino acids. This protein would
contain 64 amino acids derived from the amino terminus of the F-MuL
V gp70, including the signal peptide, 36 amino acids from a central
region of the F-MuLV env gene, and 184 amino acids that are specific
to MPL V (Fig. 1). A hydrophobicity plot of the amino acids sequence
revealed that, in addition to the 34 hydrophobic amino acids of
the gp 70 signal peptide, the MPL V -specific domain contained a
stretch of 22 uncharged amino acids. Thus, the putative MPL V env
product presents the features of a transmembrane protein comprising
an extracellular domain of 143 amino acids, a single transmembrane
domain of 22 amino acids, and a cytoplasmic domain of 119 amino
acids without consensus sequence for kinase activity [14]. Computer
analysis of the deduced amino acid sequence revealed that the MPL
V -specific sequence did not correspond to any known genes. 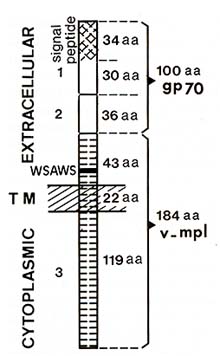
Since nonviral sequences found in the genome of acutely transforming
retroviruses derive from cellular genes and are conserved phylogenetically,
we looked for the presence of MPLV -specific sequences in genomic
DNA from different mammals. Under stringent hybridization conditions,
discrete bands were revealed in DNAs from mouse, rat, mink, dog,
cow, and human. In addition, MPLV specific probes recognized a 3.0-kb
mRNA in spleen and bone marrow from adult mice and in fetal liver
cells, but not in nonhematopoietic tissues [11 ]. Thus, taking into
consideration the biological properties of MPL V, the cellular origin
of the sequence contained in its env gene, the conservation in the
genome of mammals and the expression in normal hematopoietic tissues,
we concluded that MPL V had transduced a novel oncogene which was
designated as v-mpl. By in situ hybridization and genetic analysis
studies, chromosomal localization of the c-mpl proto-oncogene was
assigned to mouse chromosome 4 (Vigon et al., unpublished data)
and to human chromosome 1 p34 [15]. Leukemogenic Properties of MPLV In vivo studies by Wendling and coworkers have indicated that
MPL V induced a rapid suppression of growth factor requirements
for in vitro colony formation of a large spectrum of committed as
well as multipotential progenitor cells [16, 17]. The primary manifestation
of viral infection was a switch to erythropoietin (EPO) independence
of the colony forming unit-erythroid (CFU-E) population which was
complete in the spleen after 6 days of infection. A possible stimulating
effect of EPO present or secreted in the culture medium was ruled
out by the addition of neutralizing antiEPO antibodies to the culture
system. The effects of MPL V infection on the early and primitive
erythroid progenitor cells (BFU-E) was assessed in methylcellulose
serum-free cultures. It was found that well hemoglobinized pure
and mixed erythroid colonies developed without the addition of interleukin-3
or EPO. Moreover, while a majority of colonies contained erythroblasts
mixed with megakaryocytes, about 12% revealed three or more lineages
of differentiation [16]. Further in vivo studies have documented
that MPL V infection also induced the spontaneous colony formation
of myeloid progenitors, i.e., granulocyte macrophage colony-forming
cell (GMCFC) granulocyte (G)-CFC, mega karyocyte (Meg)-CFC, and
mixed CFC, probably as a result of direct infection of these progenitors
and not as a consequence of a paracrine secretion of soluble colony
stimulating factors by the accessory cells [17]. These observations
supported the conclusion that MPL V acts on various progenitors,
inducing their proliferation and terminal differentiation independently
of signals normally provided by colony stimulating factors, interleukins,
EPO, or any conditioned medium. However, formal proof that MPL V
can transform hematopoietic target cells in the absence of coinfection
with a replicating MuL V was not provided by these experiments.
We addressed this question by producing helper-free MPL V stocks
using the packaging psi-CRE cell line that produces a high titer
of infectious, nonreplicating particles but does not yield helper
virus [18]. When adult ICFW mice were intravenously given helper-free
preparations of MPL V, more than 90% of the mice were healthy 2
months after inoculation. Nevertheless, we observed that MPL V induced
a mild but transient spleen enlargement with the appearance of colonies
well visible on the spleen surface on days 5, 10, and 15 after inoculation.
Histologically, colonies were composed of erythroblasts, or erythroblasts,
granulocytes, and megakaryocytes clustered together in the splenic
red pulp. On day 25 and thereafter, these colonies disappeared,
leaving spleens with a normal aspect. In contrast, when helperfree
preparations of MPL V were injected into mice pretreated with the
aplastic drug 5- fluorouracil ( 5- FU, 150 mg/kg body weigth, 4
days before virus inoculation), all animals developed atypical MPL
V syndrome and died from overt leukemia within 2 months (Wendling
et al., unpublished data). Together these data indicate that
An area of current research in our laboratory is related to the ability of a helperfree preparation of MPL V to transform hematopoietic cells in vitro. A 2-h incubation of bone marrow cells enriched in highly dividing primitive progenitors by treatment of mice with 5- FU was sufficient to induce autonomous colony formation of about 30% of the colonyforming cells present in the preparation. Cytologically, half of these spontaneous colonies were composed of either granulocytes, megakaryocytes, or erythrocytes, while the remainders were mixed colonies of which about 20% contained three or more lineages of differentiation. Upon replating, the multilineage colonies produced secondary and tertiary mixed colonies, suggesting self-renewal [11]. The question of whether or not transformation of hematopoietic progenitors would lead to the generation of immortalized cell lines was then investigated. When marrow cells were cultured in liquid medium, it was observed that rapidly dividing nonadherent cell populations were produced in MPL V -infected cultures. After 10 to 12 days, these nonadherent populations could be transferred into fresh flasks devoid of stromal feeder layers. Cells continued to proliferate and generated permanent suspension cultures containing polymorphonuclears, megakaryocytes and erythroblasts. Upon continuous passages, the majority of the cell lines evolved towards a more restricted phenotype which remained stable over several months. Diverse immortalized megakaryocytic, myelomonocytic, erythroblastic, or mastocytic cell lines retaining the ability to differentiate could easily be obtained. Since these permanent cell lines evolved from a multipotential to a more restricted phenotype, we investigated whether they were polyclonal or monoclonal by studying proviral-cell DNAjunctions. Cultures were polyclonal 5 days after initiation. However, after 3 weeks and at a time where all cultures displayed a multipotential phenotype, one or a few major proliferating clones were detected in each cell line. Interestingly, the same clones were still found after 3 months of continuous passages when the cell lines appeared to be restricted in their differentiation potential [11]. Thus, it seems likely that MPLV induces the clonal outgrowth of a single or few transformed, probably multipotentiaI, stem cells (clonal selection), the full differentiation capabilities of which being lost along with continuous culturing (clonal evolution). The obtaining of immortalized in vitro cell lines raised the question of whether cells were tumorigenic. To approach this problem, 2 x 106 cells were subcutaneously grafted into either syngeneic or nude mice. Upon repeated assays, none of the cell lines developed tumor nodules at the site of inoculation when cells from cultures less than 4 months old were grafted. After prolonged passages (more than 7 months), 60% of the cell lines produced hematopoietic subcutaneous tumoral nodules, suggesting that additional genetic events must have occurred to reach a full malignant state.
The myeloproliferative leukemia virus isolate consists of two distinct
viral components: a replicating F-MuLV and a helper-dependent MPL
V. MPL V accounts for the rapid in vivo and in vitro transformation
of a broad spectrum of multipotential, myeloid, and erythroid progenitors
which acquire growth factor independent proliferation and differentiation.
By sequence analysis of a biologically active clone, MPL V has been
shown to be an env recombinant virus containing sequences derived
from the F-MuL V env gene and additional nonviral cellular sequences.
These nonviral sequences are conserved in various mammals and are
expressed in hemopoietic tissues from normal mice. MPLV was thus
generated by transduction of an oncogene (v-mpl) in the envelope
region of an F -MuLV genome. v-mpl does not correspond to any known
gene, but the putative MPL V env fusion product has the features
of a transmembrane protein with the Nterminal signal sequence of
the F-MuLV gp70 directing the polypeptide across the membrane and
a single transmembrane domain. Interestingly, the extracellular
domain of v-mpl possesses, 13 amino acids upstream to the membranespanning
domain, the amino acid sequence WSXWS, highly conserved in all cy
to kine receptors that make up the hematopoietin receptor superfamily
[19]. In addition, a significant number of conserved amino acids
were found when the extracellular domain ofv-mplwas aligned with
that of the IL-2ß, IL-3, IL-4, IL-6, IL- 7, GM-CSF, G-CSF, and EPO
receptors [11]. Since the N-terminal part of the fusion protein
consists of F -M uL V derived sequences, it is not yet known whether
the c-mpl proto-oncogene product would contain the highly conserved
cysteine residues characteristically found in the ligand-binding
domain of each of these receptors [ 19] .Nevertheless, with regard
to the general features of v-mpr, it is tempting to speculate that
MPL V has transduced a truncated form of a putative cytokine receptor.
Cloning of the protooncogene cDNA is currently underway in our laboratory
to allow further comparison. A major focus of future research will
be to understand the mechanism by which this viral oncogene can
short-circuit the growth-regulatory signals delivered by the binding
of various hematopoietic growth factors to their specific receptors.
This requires further studies on the mechanism of signal transduction
by MPL V and by other receptors of the same family. Acknowledgments.
|