|
1 Shemyakin Institute of Bioorganic Chemistry,
USSR Academy of Sciences, Moscow, USSR.
Muramyl pep tides (MPs) are known to influence greatly the immune
response [1]. They are active as adjuvants, induce nonspecific resistance
to certain viral and bacterial infections, and in some cases are
active against various tumors. Many studies attempting to elucidate
the mechanism of their biological activity have been carried out.
However, the molecular basis of MP's effects on the immune system
is still unclear . The immune network includes at least three circuits:
idiotype-antiidiotype interactions [2], the cytokine network [3],
and the regulation of receptor expression on immunocompetent cells.
MPs are known to induce cytokine production, and so clearly interfere
with the cy to kine cascade [4]. They also influence idiotype network,
as they increase production of immunoglobulin. Little if anything
is known about their effect on the expression of cell membrane molecules
[5]. We approached this subject by studying oligosaccharide-containing
MPs synthesized in our institute by T. Andronova, E. Makarov, and
V. Ivanov. Macrophages are known to be the primary target for MPs
[1]. As these cells function as antigen-presenting cells, we evaluated
the effect of N-acetylglucosaminyl-ß 1-4-N-acetylmuramyl-L-alanylD-isoglutamine
(GMDP, Fig. 1) and its analogs on major histocompatibility complex
(MHC) class II antigen expression, as these glycoproteins are crucial
for recognition of antigens by T cells [6]. BALB/c mice peritoneal
macrophages were used. The number of Iapositive cells was estimated
by flow cytometry after labelling cells with biotinylated anti-Ia
monoclonal antibodies and fluorescein isothiocyanate (FITC)labelled
avidin.
To summarize briefly the results obtained [7], it was found that:
1) Incubation of macrophages with GMDP in vitro caused a dosedependent
increase in the Ia-positive cell number and fluorescence intensity.
la expression peaked at 18 h and persisted for at least the next
30 h.
2) This effect was also observed in vivo upon injection of GMDP
into the peritoneal cavity of mice. '
3) Biologically active MPs, with few exceptions, also possessed
Ia-inducing activity; nonactive compounds were always inactive in
this assay.
4) MPs directly affected macrophage. This was evident from the increase
in Ia-antigen expression on myelomonocytic leukemia cells (WEHI-3).
The effect could not be attributed to intermediate formation of
tumor necrosis factor alfa (TNF-alfa ), the known la inducer, as
we failed to find TNF -alfa in WEHI-3 culture medium. At present,
the involvement of other interleukins cannot be excluded.
5) The observed effect was not species specific: not only murine
peritoneal macrophages but human monocytes as well could be induced
to express MHC class II (HLA-DR) antigens, though the magnitude
of the effect was lower. The maximal HLA- D R expression was observed
at 0.1-1 µg GMDP/ml.
6) Finally, besides MHC class II antigens, the expression of interleukin-2
(IL-2) receptors was induced by GMDP. Taking in account that IL-2
was shown to increase the cytotoxicity of macrophages against tumor
targets, we assume that this mechanism might be involved in the
antitumor activity of GMDP.
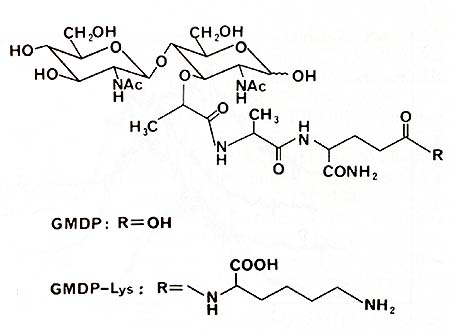
Fig.1. N-Acctylglucosaminyl-ß1-4-Naceglucosaminyl-ß1-4-N
acetylmuramyl-L-alanyl-D - isoglutaminc (GMDP) and GMDP-Lys
It was shown previously that MPs have no direct cytotoxic effect
on tumor cells; rather, they kill tumor cells by activating the
immune system [8]. We assumed that there might be another effect,
namely an increase in expression of tumorassociated antigens and
MHC antigens on tumor cells, resulting in their being better recognized
by immunocompetent cells. Human lung adenocarcinoma cells (RL-4)
and colon adenocarcinoma cells (WiDr) were used as tumor targets.
These cells are known to express various levels of carcinoembrionic
antigen (CEA) as tumor-associated marker. The expression of CEA
and RL-4 cells was monitored by labelling the cells with FITC-anti-CEA
monoclonal antibodies followed by flow cytometric enumeration of
labelled cells. RL-4 cells cultured without stimulant expressed
only minute amounts of CEA. Incubation of RL-4 cells (3 x 105) with
GMDP resulted in a dose-dependent increase in CEA expression with
maximal expression at 10 µg/ml (Fig.2). The number of CEA-positive
cells peaked at 18-24 h. An additive effect was observed upon combined
treatment ofRL-4 cells with GMDP and interferon-gamma (IFN-gamma).
The treatment of RL-4 cells with GMDP also resulted in increased
expression of HLA-DR antigens. For WiDr cells an increase in CEAexpression
after 48 h incubation was observed as well. In this case, flow cytometry
could not be used due to clumping of cells. The CEA expression by
WiDr cells was monitored by cell enzyme-linked immunosorbent assay
(ELISA). Whether the above-mentioned effects have functional implications
regarding the recognition of tumor cells by the immune system remains
to be studied. Another goal of our study was to identify the MP-binding
molecules on responding cells, as the existence and location of
specific cellular MP receptors were controversial [9,10,11]. To
address this question we used two approaches. The first approach
was based on direct staining of MP-binding cells with FITClabelled
GMDP-Lys: the FITC group was attached to the epsilon-amino group
of lysine (see Fig. 1). Fluorescence-activated cell sorting (F ACS)
analysis of intact murine peritoneal macrophages and WEHI-3 cells
showed no specific binding, but when the cell membrane was first
fixed with paraformaldehyde and permeabilized with ß-octylglucoside
and then treated with GMDP-Lys-FITC,
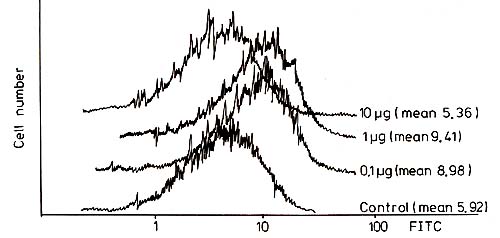
Fig.2. Effect of incubation with GMDP on CEA expression
by RL-4 cells
binding to macrophages and WEHI-3 cells was observed. This binding
could be inhibited effectively by GMDP or GMDP-Lys. In contrast,
the tripeptide Ala-Gln-Lys was rather ineffective as an inhibitor
. Thus, the GMDP-binding molecule seems to be located inside macrophages.
The number of specific binding sites on WEHI-3 cells estimated by
comparison with fluorescein-labelled beads of known molecular composition
was in the range of 6 x 10 high 4, though the total number of binding
sites was much higher (2.2 x 10 high 5). These values are lower
than those obtained for rabbit bronchoalveolar lavage cells [12].
The second approach was based on evaluation of fluorescence polarization
of GMDP-Lys-FITC upon binding to cells. Similarly to F ACS analysis,
only for permeabilized cells was specific binding observed, detected
by a change in fluorescence polarization. The ability of GMDP and
GMDP-Lys to compete with fluorescent congeners for binding sites
suggested specificity of binding. The number of specific binding
sites per cell calculated from these data was in the range of 4-5
x 10 high 4 and agreed reasonably well with data obtained by F ACS
analysis. The Scatchard plot suggested the presence of two populations
of binding sites with Kd 2 x 10 high -8 M and 5 x 10 high -7 M (Fig.
3). The above findings are consistent with intracellular MP-binding
molecules. Thus, to display immunomodulatory activity MP must be
internalized by macrophage. The binding of GMDP to a receptor molecule
results in initiation of biosynthesis or enhanced biosynthesis of
certain proteins, including cytokines and cell membrane molecules.
The changes in surface antigen expression influence the magnitude
of the immune response.
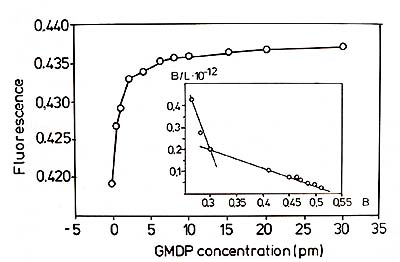
Fig.3. Binding of GMDP
Acknowledgments.
The authors would like to thank Dr. J. Shively (Los Angeles, USA)
for kindly providing with WiDr cells and anti-CEA antibodies, Dr.
G. Hammerling (Heidelberg, FRG) for anti-Ia producing hybridoma
(B17-123), Dr. H. Wagner (Munich, FRG) for antiIL2-receptor monoclonal
antibody, and Dr. E. Revazova for RL-4 cells.
References
1. Adam A, Petit JF, Lefrancier P, Lederer E (1981) Molec Cell
Biochern 41 :27-47
2. Perelson AS (1989) Immunol Rev 10: 5 36
3. Balkwill FR, Burke F (1989) Immunol Today 10:299-304
4. Abehsira-Amar 0, Damais C, Parant M, Chedid L (1985) J Immuno1134:365-368
5. Behbehani K, Beller Dl, Unanue ER (1985) J Immuno1134:2047-2049
6. Weaver CT, Unanue ER (1990) Immunol Today 11 :49-55
7. Nesmeyanov V A, Khaidukov SV, Koma leva RL, Andronova TM, Ivanov
VT (1990) Biomed Sci 1: 151-154
8. Ivanov VT, Andronova TM, Bezrukov MV, Rar V A, Makarov EA, Kozmin
SA, Astapova MV, Barkova TI, Nesmeyanov VA (1987) Pure Appl Chern
59(3):317-324
9. Leclerc C, Chedid L (1986) Int J Immuno ther 11 : 109 -114
10. Silverman DHS, Krueger JM, Karnovsky ML (1986) J Immuno1136:2195-2201
11. Tenu JP, Adam A, Souvannavong V, Yapo A, Petit JF, Douglas
K (1989) Int J Immunopharmaco111 :653-661
12. Richerson HB, Adams P, Upadrashta B, Iwai Y, Barfknecht C (1988)
J Allergy Clin Immunol 81: 171
|


