|
Institut für Virologie, Zentrum für Hygiene. Universität Freiburg. Hermann-Herder-Str. I I. 7800 Freiburg, Federal Republic of Germany In recent years it became increasingly clear that infection of
the human host by a number of viruses results in a probably life-long
persistence of these agents within specific cells (reviewed e.g.
zur Hausen, 1977a). Reactivation of persisting genomes is a well-known
phenomenon in individuals infected by herpes group viruses (herpes
simplex virus, varicella zoster virus) where neural cells have been
identified as site of virus persistence. Infection with Epstein-Barr
virus, the causative agent of infectious mononucleosis, leads to
viral genome persistence in certain B-lymphocytes. They can be isolated
and are readily propagated in tissue culture even years or decades
after primary infection (reviewed by zur Hausen, 1975). Transmission
of cytomegalovirus infections by blood transfusions has been frequently
recorded, even from donors who acquired the virus several years
earlier . Besides herpes group viruses, however, it became more
and more obvious that a number of additional agents may persist
continuously within the human host. Adeno-viruses seem to be able
to persist for long periods within adenoids and specific cells of
the tonsils (reviewed by Green, 1970), hepatitis B virus seems to
be a candidate for extremely long periods of persistence (Zuckerman,
1975) and the 2 human polyoma-like viruses, BK and JC, apparently
remain latent for life-time. Reactivation of the latter appears
to be mediated by immunosuppression and may result in excretion
of large quantities of viral particles in the urine of affected
patients. The papilloma- or wart viruses represent additional candidates
for longtime persistence (zur Hausen, 1977 b). The observations
cited above permit the statement that every individual, increasing
with life-time, is exposed correspondingly to viral infections which
result in an increasing "burden" of persisting genomes within certain
cells. Although it is presently impossible to predict the actual
percentage of cells being affected by viral genome persistence at
a given age, it is probablyjustified to assume a substantial number
in every cell compartment of human adults. This can be considered
as an epigenetic potential which may remain genetically silent in
most instances. It is a remarkable feature of a substantial number
of these persisting agents that many of them are oncogenic when
inoculated into non-natural hosts: 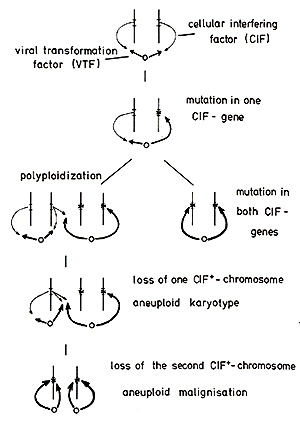
the controlling CIF alleles. The postulation of a balanced control
by CIF of the effector gene(s) (zur Hausen, 1977a) predicts an enhanced
growth potential of effector-positive cells carrying am utation
in one CIF gene of the allelic set. This would result in the outgrowth
of monoclonal, phenotypically normal cells and could explain observations
by Fialkow (see this volume) in patients with chronic myelogenous
leukemia and erythrocythemia vera, revealing not only monoclonality
in the respective tumor cells but also in normal cells derived from
the same steillline. Mutations in both sets of CIF genes of an effector-containing
cell should be an extremely rare event if we consider the spontaneous
mutation rate for a specific gene (Spandidos and Siminovitch, 1978;
Barret and Ts'o, 1978). They would result in immediate "illalignisation"
of a diploid tumor cell. Such events, however, would be 5oillewhat
facilitated by selective growth advantage of cells carrying a mutation
in one CIF allele or by specific integration of viral DNA into these
genes or specific mutagenisation due to viral or other agents (zur
Hausen, 1967). Polyploidization should enhance the progression to
malignancy in some subsequent steps. Polyploid cells show an increased
tendency to loss of individual chromosomes and it should be statistically
predictable how many cell divisions are required before the loss
of both functioning alleles results in an aneuploid tumor cell.
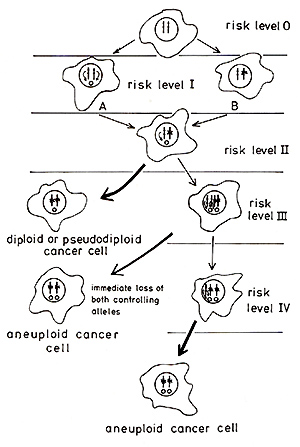 Fig.2 The model permits the staging of risk levels for malignisation
ofindividual cells. This is attempted in Fig. 2. An effector-free
normal cell would be at risk level 0. It is questionable whether
such cells exist in reality since genes of endogenous (vertically
transmitted) viruses and possibly also genes of cellular origin
may possess effector properties. It is possible, however, that the
difficulties in transforming human cells in tissue culture by chemical
and physical carcinogens (initiators) when compared to rodent cells,
may result from the lack of effectors or their reduced number in
comparison to e.g. mouse cells. The model of carcinogenesis described
here conveniently explains some of the prevalent features of cancer
cells:
1. Albrecht T., Rapp, F.: Malignant transformation of hamster embryo fibroblasts following exposure to ultraviolet-irradiated human cytomegalovirus. Virology 55,53-61 ( 1973 ) 2. Barrett, J. C., Ts'o, P. 0.: Relationship between somatic mutation and neoplastic transformation. Proc. Natl. Acad. Sci. USA 75,3297-3301 (1978) 3. Burnet F. M.: The concept of immunological surveillance. Prog. Exp. Tumor Res. 13, 1-28 (1970) 4. Duff, R.. Rapp, F.: Properties of hamster embryo fibroblasts transformed in vitro after exposure to ultraviolet-irradiated herpes simplex virus types. J. Virol. 8,469-477 (1971) 5. Epstein, M. A., Hunt R. D., Rabin, H.: Pilot experiments with EBV in owl monkey (Aotustrivirgatus). I. Reticuloproliferative disease in an inoculated animal. In t. J. Cancer 12,309-318 (1973) 6. Green, H.: Oncogenic viruses. Ann. Rev. Biochem. 39,701-756 (1970) 7. London, W.T., Houff, S.A., Madden, D.L., Fucillo, D.A., Gravell. M.. Wallen. W.C.. Palmer. A. E., Sever, J. L., Padgett B. L., Walker, D. L., Zu Rhein, G. M.. Ohashi. T.: Brain tumors in owl monkeys inoculated with a human polyomavirus (JC virus). Science 201, 1246-1249 (1978) 8. Shope, T., Dechairo, D., Miller, G.: Malignant lymphoma in cottontop marmosets following inoculation ofEpstein-Barr virus. Proc. Natl. Acad. Sci. USA 70,2487-2491 ( 1973) 9. Spandidos, D. A., Simonovitch, L.: The relationship between transformation and somatic mutation in human and Chinese hamster cells. Cel113, 651-662 (1978). 10. Stanbridge, E.J., Wilkinson, J.: Analysis of malignancy in human cells: Malignant and trans formed phenotypes are under separate genetic control. Proc. Natl. Acad. Sci. USA 75, 1466 to 1469(1978) 11. Werner, J., Wolf. H., Apodaca, J., zur Hausen, H.: Lymphoproliferative disease in a cotton top marmoset after inoculation with infectious mononucleosis-derived Epstein-Barr virus. Int. J. Cancer 15, 1000-1008 (1975) 12. Zuckerman, H.J.: Human viral hepatitis. Amsterdam: North Holland Publ. Comp. 1975 13. zur Hausen, H.: Induction of specific chromosomal aberrations by adenovirus type 12 in human embryonic kidney cells. J. Virol.1, 1174-1185 (1967) 14. zur Hausen, H.: Oncogenic herpesviruses. Biophys. Biochem. Acta 417,25-53 (1975) 15. zur Hausen, H.: Cell-virus gene balance hypothesis of carcinogenesis. Behring Inst. Mitt. 61, 23-30(1977a) 16. zur Hausen, H.: Human papilloma viruses and their possible role in squamous cell carcino mas. Current Top. Microbiol. Immunol. 78,1-30 (1977b) 17. zur Hausen. H., O'Neill, F.J., Freese. U.K., Hecker. E.: Persisting oncogenic herpesvirus in duced by the tumor promoter TPA. Nature 272,373-375 ( 1978) 18. zur Hausen, H., Bornkamm, G.W., Schmidt R., Hecker. E.: Tumor initiators and promoters in the induction ofEpstein-Barrvirus. Proc. Natl. Acad. Sci. USA76, 782-785 (1979) |