|
The James Ewing Laboratory of Developmental Hematopoicsis,
Memorial Sloan-Kettering Cancer Center, New York, NY 10021, USA
* Supported by grants CA 20194, CA 32156, and CA 31780 from the
National Cancer Institute; American Cancer Society Grant CH-3K,
and the Gar Reichman Foundation.
A. Introduction
The Frederick Stohlman Memorial Lectures have, over the years,
ref1ectcd the progression of leukemia research in areas initially
thought to be as diverse as retrovirology /oncogenes, chemotherapy
/immunotherapy, and hematopoietic growth factors. At the Seventh
Wilsede Meeting we heard how convergent these areas were. The polypeptide
growth factors which affect Iymphohematopoietic ce1l proliferation
and differentiation have expanded to at least 11 distinct gene products
with pleiotropic and overlapping functions (G-, GM-, M-CSF, erythropoietin,
and interleukins 1- 7 [1 ]. The early characterization of hematopoietic
growth factors was based on their ability to stimulate the clonal
proliferation of human bone marrow progenitor ce1ls. In addition,
these factors were shown to activate a variety of functions of mature
lymphoid and hematopoietic ce1ls. The purification, characterization,
and gene cloning of human G-CSF fo1lowed shortly after the equivalent
characterization of human GM-CSf [2, 3]. With the availability of
recombinant G-CSF in quantities sufficient for in vivo evaluation,
its action on hematopoietic function was assessed in normal murine
and primate systems, and fo1lowing chemotherapy or radiation-induced
myelosuppression [1, 4, 5]. Efficacy was rapidly demonstrated and
opened the way to clinical trials in human bone marrow suppression
or failure systems [6 8]. Without reviewing this area in detail,
1 wish to touch on various aspects that i1lustrate the utility of
G-CSF in various pathophysiological situations.
B. Hematopoietic Role of G-CSF
I. G-CSF Treatment of Mice Receiving Cyclophosphamide, Myleran,
or 5-rluorouracil
In order to investigate the potential of G-CSF in preventing episodes
of neutropenia fo1lowing high-dose chemotherapy with cyclophosphamide
(CY), C3H/ HeJ mice were suhject to weekly injections of 200 mg/kg
CY intraperitonea1ly followed by G-CSF therapy (1.75 g x 2 daily)
beginning 2 h after CY treatment and finished 48 h before the second
cycle of CY. As shown in Fig. 1, this protocol prevented the subscquent
nadirs of neutrophil counts between 4 and 5 days after CY treatment,
and in a1l ten cycles of CY therapy G-CSF ahrogated the neutrophil
nadirs. The mice receiving CY alone had a substantial mortality
evident by the eighth cycle of treatment. In eight cyc1es of CY
treatment over the course of 70 days the G-CSF-treated animals were
neutropenic (1000 ANC/mm3) for only 3 days, whereas thc untreatcd
animals were neutropenic for 24 days. The time to recovery of absolute
neutrophil counts (ANCs) to control levels was also substantially
affected, with the non-G-CSF-treated groups requiring an additional16
days for recovery. Neutropenia at levels
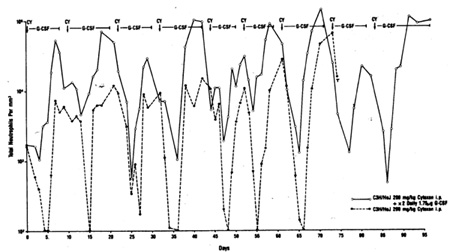
Fig. I. Absolute neutrophil counts in the peripheral blood
of C3H/HeJ mice treated with repeated dosed or 200 mg/kg cyclophosphamide
(CY) administered at 7- to 9-day intervals over 3 months. Control
mice (broken line) received saline alone; experimental mice (continuous
line) received 1.75 mg rhG-CSF twice daily intraperitoneally beginning
2 h following CY and terminating 48 h prior to the next cycle of CY.
Three mice per group. Note the log scale for ANC/mmł
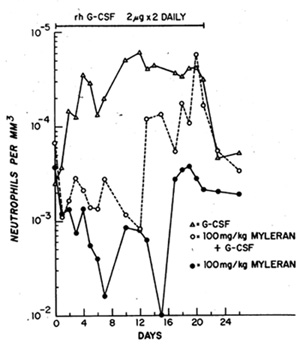
Fig.2. Absolute neutrophil counts in the peripheral blood
of C3H/HcJ mice administered 100 mg/kg myleran orally by intubation
with or without twice daily i.p. injections of rh-G-CSF at 2 µg/mouse
for 21 days. Control animals received corn oil alone with G-CSF.
Four mice per group. Note the log scale
of 500 ANC/mm3 indicated an even more significant difference, with
such low levels observed on only 1 day out of 70 in the G-CSF +
CY -treated mice, in contrast to 15 days in the CY-treated group.
This cyclophosphamide model allowed us to investigate the timing
of initiation of G-CSF therapy following a single injection of a
high dose of CY. Administration of G-CSF coincident with CY treatment
resulted in an improvernent in recovery of neutrophil counts, but
this was substantially less than observed when GCSF was administered
2 h after CY therapy. Administration of G-CSF 24 and 48 h after
CY was less effective than after 2 h, indicating that treatment
early in the course of chemotherapy is likely to be more effective.
Myleran (MY), administered orally (100 mg/kg), was also associated
with a significant reduction in neutrophil counts and in bone marrow
progenitor populations. G-CSF therapy initiated within 6 h of MY
treatment led to an accelerated recovery of neutrophils so that
by 1 week the neutrophil counts had returned to normal values, whereas
the MY -treated controls rernained profoundly neutropenic (ANC 200/rnm3)
(Fig. 2). By 14 days MY + G-CSF-treated animals exhibited a neutrophilleukocytosis
of between 104 and 2 x 104/rnm3 at times when the MY control animals
had 100 neutrophils/rnm3. Treatment with 5-fluorouracil (5-FU 150
mg/kg) led to profound myelosuppression in three different strains
of mice tested. In C3H/HeJ mice, recovery of neutrophil counts to
control values was delayed for 12-14 days. Administration of G-CSF
accelerated recovery of neutrophil counts to normal values within
8 days [5].
II. Clinical Trials of G-CSF Following Chemotherapy
The use of rh-G-CSF in clinical trials has demonstrated efficacy
in promoting regeneration of functioning neutrophil granulocytes.
Our first clinical trial involved the administration of G-CSF in
a phase 1/1I study of patients receiving MVAC (methotrcxate, vinblastinc,
doxarubicin, and cisplatin) in 22 patients with transitional ceIl
carcinoma of the uroepitheliurn [6]. In all patients a specific
dose-dependent increase in ANC was seen to levels of up to 12-fold
above normal at the higher levels of G-CSF administration. At these
high doses (10-60 g/ kg) a tenfold increase in monocytes, but not
in other hernatopoietic lineages, was evident. Treatment after chernotherapy
significantly reduced the number of days on which antibiotics were
used. This resulted in a significant increase in the number of patients
qualified to receive planned chernotherapy on day 14 of the treatment
cycle (100% vs. 29% ). In addition, the incidence of mucositis significantly
decreased. Investigators at other centers have confirmed these observations.
In patients with advanced small cell lung cancer receiving high-dose
chemotherapy, repeated every 3 weeks, G-CSF was given to each patient
for 14 days on alternate cycles of chemotherapy. This resulted in
the reduction of the period of absolute neutropenia with return
to normal or supranormallevels of ANC within 2 weeks [8]. This therapy
also drastically reduced episodes of severe infections observed
during these cycles of chemotherapy .In advanced malignancies treated
with melphalan, neutropenia was significantly reduced even at dose
levels of 1 or 3µg/kg G-CSF. Preliminary clinical studies
have also demonstrated that G-CSF hastens granulocyte recovery in
Hodgkin's disease after high-dose chernotherapy and autologous bone
marrow transplantation [9]. In all of these studies no significant
dose-Iirniting toxicities have been observed to date.
III. In Vivo Studies of G-CSF in Congenital and Idiopathic Neutropenia
Cyclic neutropenia is an inherited disease of man and gray collie
dogs, characterized by regular oscillations of the number of peripheral
blood cells and of bone marrow progenitor cells. Cycling of se rum
or urine CSF levels has also been reported. However, induction or
cure of cyclic neutropcnia by bone marrow transplantation has suggested
that this disorder is a disease of pluripotential hematopoietic
stern cells. It should be noted that the hematopoietic stem cell
itself can generate cells (macrophages, T cells ) capable of hematopoietic
growth [actor production and thus it is not possible to exclude
intrinsic growth factor production defects in the pathophysiology
of cyclic neutropenia. In the dog model we have demonstrated that
daily administration 0f rh-G-CSF (5 µg/kg x 2 daily [or 30
days) caused an immediate (within 12 h) and persistent leukocytosis
( > 40 000 W HC) in both cyclic and normal dogs due to a ten [old
increase in the numbers 0[ circulating neutrophils and monocytes
[10]. This therapy eliminated two predicted neutropenic episodes
and suppressed the cycling of CFU-GM in the bone marrow. Comparable
therapy with rh-GM-CSF induces a monocytosis and neutrophilia in
normal dogs but did not eliminate the recurrent neutropenia in cyclic
dogs. Analysis 0[ the serum levels of CSF indicated a striking periodicity
with peak levels of G-CSF coinciding with the peaks of monocytes
and the nadirs of neutrophils. The continuing cycling of CSF levels
was also observed even when animals showed a major neutrophil leukocytosis
under exogenous GCSF therapy. A disorder of neutrophil regulation
is also seen in chronic idiopathic neutropenia in which the peripheral
neutrophil count is reduced to 2000 cells/mm3 for prolonged periods,
with maturation arrest of neutrophil granulocyte precursors in the
bone marrow. Other hematopoietic cell counts are usually normal
and antineutrophil antibodies are absent. Clinically, these patients
experience recurrent episodes of life-threatening infections, ulcers
of the mucus membrane, and periodontal disease. We have had the
opportunity of studying a number of patients with this disorder
utilizing primary clonogcnic assay and in vitro suspension cultures
of patient bone marrow cells [7]. The incidence of CFU-GM observed
in primary bone marrow culture was always in the high range of normal
with respect to each species of CSF (G-, GM-, M-, and IL-3), indicating
that the neutropenia was not attributed to a shortage of appropriate
myeloid progenitors. furthermore, morphological studies showed normal
neutrophil maturation within the colonies developing in the presence
of G-, GM-CSF, and IL-3, with eosinophil maturation and macrophage
development a significant feature with GM-CSF stimulation. In suspension
cultures GCSF was particularly effective in generating mature segmented
neutrophils which were absent in input bone marrow, and did not
develop spontaneously in over 3 weeks of culture. With the addition
of G-CSF, myelocytes expanded in the 1st week 0[ culture. By the
2nd and 3rd week high levels of production of segmented, functionally
normal neutrophils were found. Based upon these in vitro studies,
patients were treated with subcutaneous G-CSF on a daily continuous
basis. In the first patient studied, the neutrophil count rose rapidly,
and by 20 days the patient's ANC was > 1000 cells/mmł and plateaued
in the range of 2000-3000/mmł with evidence of a 40-day cycle (observed
even when the dose 0[ G-CSF was increased). The neutrophils were
functionally normal and the patient has remained on this continuous
G-CSF treatment without any notable toxic side effects [7]. A more
severe form of neutropenia is found in patients with Kostmann's
syndrome (congenital agranulocytosis). In this disorder, marked
by sevcre neutropenia and maturatjon arrest at the myelocyte level,
five patients received GCSF therapy, and the neutrophils increased
from 0% -1% to 10%-72% with clinical resolution of preexisting infections
[11].
C. Hematopoietie Role of Interleukin-1
Interleukin-1 ( I L-1 ) was first identified as an endogenous pyrogen,
produced by macrophages following activation by endotoxin. Subsequent
studies implicated IL-1 as a mediator of a variety of inflammatory
phenomena involving production of cascades of cytokines and cyclooxagenase
products [12]. Various lymphokines are released by the interaction
of I L-1 with T cells, and IL-1 induces expression of G-, GM-, and
MCSF by a direct action on stroma] cells such as fibroblasts and
endothelial cells, both in vitro and in vivo [13 -15]. Recent studies
have revealed a more direct role for IL-1 in hematopoiesis, involving
an interaction with the early hematopoietic stern cell [5,16-18].
I. Interleukin-1 Identity with Hemopoietin 1/Synergistic Activity
Early studies involving mice treated with 5-fluorouracil (5-FU)
suggested the necessity of a synergistic interaction between two
factors, one being a direct hematopoietic colony stimulus such as
MCSF or IL-3, and the other, which lacked direct colony-stimulating
activity, was shown to synergize with the preceding species of CSFs
[19]. Syncrgistic activity was identified in conditioned media or
extracts of various human tissues and was also identified and purified
to homogeneity from the human bladder cancer cell line, 5637 [20].
This latter cellline had also been used to identify, purify, and
clone G- and GM-CSF [2, 3, 21]. The constitutive expression of hemopoietin
1 (H-1), which was shown to be a 17000kd protein [20], led us to
attempt to purify, sequence, and clone this molecule, using similar
strategies to those that we had employed to clone the G-CSF gene
from 5637 cells. The identity of [L-1 and H-11 synergistic activity
was established on the basis of the following factors :
1. The bladder cancer cells evidently produced high levels of IL-1
active in the thymocyte comitogenesis assay.
2. The synergistic activity and I L-1 could be copurified using
various protein purification procedures.
3. Abundant levels of IL-1-alpha and -beta mRNA could be detected
in the 5637 cells -comparable to levels in activated macrophages.
4. F'ractions of in RNA from 5637 expressed in oocytcs resulted
in intracellular production of hematopoietic synergistic activity
and thymocyte comitogenesis factor production.
5. The synergistic activity and thymocyte comitogenesis activity
were completely neutralized by monoclonal and polyclonal antibodies
to IL-1. 6. rhlL-1 alpha and beta at 0.110 U/ml synergized with
G-, GM- M-CSF, and IL-3 in stimulating high proliferative potential
(HPP)-CFU in clonogenic assays of 5-FU-treated murine bone marrow
[1, 5, 16-18].
IL-1 has a direct effect upon hematopoietic stem cells in addition
to its ability to elicit production of various CSF species by accessory
cell populations within hematopoietic tissues. In our original studies
it was not possible to conclude that I L-1 was acting directly on
early stern cells since accessory cell populations were not depleted
from the target bone marrow cell population. In more recent studies
we have established a linear doseresponse relationship between the
numbers of HPP-CFU, and the number of bone marrow cells plated,
with a highly significant correlation (r = 0.97) indicative of a
single-hit phenomenon.
II. Action ofIL-1 in Short-term Marrow Suspension Culture (Delta
Assay)
The rationale behind the delta assay is to demonstrate the ability
of hematopoictic growth factors to promote the survival, recruitment,
or expansion of stern cells and/or progenitor cells in relatively
short term suspension culture systems. As originally developed,
we utilized bone marrow from mice that had been treated with 5-ru
for 24 hand then subjected to a 4- to 7-day suspension culture in
the
Table I. Interleukin-1 and CSF-induced amplification of
CFU-GM in 7-day
suspension cultures of 25 x 100.000 murine bone marrow cells obtained
24 h post 5-fluorouracil treatment
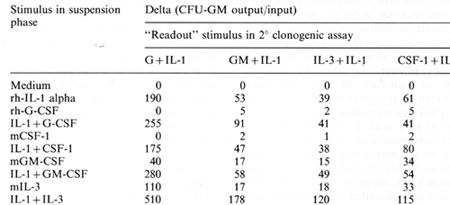
Femoral bone marrow cells taken from B6D2F1 mice 24 h after a
single i.v. injectiol1 of 5-nuorouracil (150 mg/kg) were incubated
at 25 x 100.000 cells/ml in Iscove's modified Dulbecco's medium
with 20% fetal calf serum il1 24-well cluster plates containing
100 units rh-1 L-I alpha. or 2000 u/ml rh-G-CSF; 1000 u/ml m-GM-CSF
(purified from muril1e post-endotoxil1 lung CM) 200 u m-IL-3 (purified
from WEHI-3 cell line CM), or 1000 u CSF-1 (purified from L-cell
CM). After 7 days of incubation, cells were recovered and assayed
for CFU-GM in agarose cultures stimulated by the various CSF species
alone or in combination with IL-1. The delta valuc (CFU-GM output/input)
was calculated on a recovery from triplicate clonal assays from
triplicate suspension cultures
presence of I L-1 alone, CSFs alone, or combinations of IL-1 with
various CSF species [5, 16]. At the end of the suspension culture
phase, total cellularity and morphology was determined, and cells
were cloned in semisolid culture, again in the presence of IL-1
alone, CSFs alone, or combinations of I L-l and CSFs. Table 1 shows
that IL-l caused an expansion of the numbers of CFU-GM recovered
after 7 days of culture. Neither M-CSF nor G-CSF alone supported
survival or expansion of CFU-GM; however, the combination of IL-l
and CSFs demonstrated additivc or synergistic effects on the expansion
of thcsc progenitors. GM-CSF or IL-3 alone caused some expansion
of progenitor populations, but again the combination with lL-1 evidenced
synergtsm. Human systems based on in vitro purging with 4-hydroperoxcyclophosphamide
(4HC) and positive selection by "panning" with MY10 monoclonal anti
body for CD34 + cells followed by 7-day suspension culture also
demonstrated synergistic interactions between 1 L-1 and various
CSF species. The most dramatic effect was observed with the combination
of IL-1 and IL-3, where an up to 85-fold increase in progenitor
cells was noted following 7-day suspension culture (Table 2). In
contrast to the murine system, synergism between IL-1 and CSF-1
was not evident.
III. In Vivo Interaction Between IL-1 and G-CSF in mice Treated
with 5-FU
In mice trated with 5-FU, G-CSF administration restores neutrophil
counts to normal values some 5-6 days earlier than in mice not receiving
the factor, but aperiod of profound neutropenia is still observed
[5]. Administration of IL-l alone, giving postchemotherapy for 410
days twice daily at doses of 0.2 µg/ mouse per day, reduced
the severity of
Table 2. Interleukin-1 and CSF-induced amplification of
CFU-GM in 7-day
suspension cultures of 25 x 100.000 4-HC-treated, CD34 + human bone
marrow cells
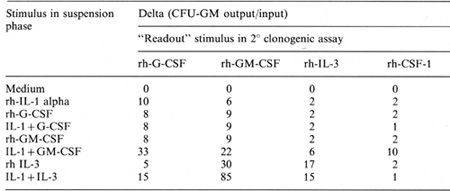
Bone marrow cells, obtained with informed consent from normal
volunteers, were separated over Ficoll-Hypaque, subjected to plastic
adherence, and incubated for 30 min with 100 µM 4-hydropcroxycyclophosphamide
(4-HC). Cells were then treated with antimyl monoclonal antibody
(anti-HPCA-1 Beckon Dickinson, Mountain View Ca), on ice for 45
min, washed. al1d incubated for 1 h at 4 Celsius on bacteriological-grade
plastic petri dishes previously coated with goat anti-mouse IgCJ.
Adherent CD34 + cells were harvested by vigorous pipcttil1g. 25
x 100.000 harvested cells/well were il1cubated in lscove's moditied
Dulbecco's medium (IMDM) plus 20%., fetal calf serum in 24-well
cluster plates containing test stimuli. In suspension phase, stimuli
were 10 ng/ml rh-G-CSF, rh-GM-CSF, rh-IL-3 (Amgen), rh CSF-1 (Cetus),
and rh-IL-1 alpha (Roche). 4-HC purged, CD34 + marrow cells were
plated at 2 x 10.000 cells/ml in semisolid agarose culture in the
presence or 10 ng/ml GM-CSF, G-CSF, CSF-1, or IL-3 alone or in combination
with IL-I both pre- and postsuspension culture. Colony formation
was assessed after 12 days. The delta value (CFU-GM output/input)
was calculated on recovery from triplicate clonal assays from duplicate
suspension cultures
the neutrophil nadir, and accelerated the recovery of the neutrophil
count to an extent greater than observed with G-CSF alone. The combination
of G-CSF and IL-1 administered after 5-FU therapy also resulted
in accelerated hematopoictic reconstitution, although the results
were additive rather than synergistic [5]. Analysis of total hematopoietic
cell reconstitution in %-FU-treated mice also showed the efficacy
of I L-1 therapy, or IL-l plus G-CSF therapy, in accelerating total
recovery of erythroid as weil as granulocytic elements in the marrow,
spleen, and blood . The potential of combination biotherapy as an
effective means or accelerating hemopoietic cell differentiation
raises the issue of whether premature exhaustion of the stern cell
and progenitor cell population may occur. Measurement of colony
forming units (cru-s, CFU-c, BFU-c, CFU-GEMM, CFU-Meg) on an incidence
basis proved to be misleading because of the redistribution or hematopoietic
precursors in regenerating murine tissues. By quantitation of total
numbers of these cell populations in marrow, spleen, blood, and
other tissues, we demonstrated that there was an absolute increase
in the recovery of all of these populations in 5-FU-treated mice
exposed to both I L-1 alone and I L-1 plus G-CSF. The results indicate
that the mechanism of action of these cytokines, both alone or in
combination, is not simply mediated by accelerated differentiation,
but involves an absolute expansion of the most primitive stern cell
populations that can be measured, e. g., by the day 12 murine spleen
colony (CFU-s) assay, or the high proliferative potential in vitro
colony assay requiring IL-1 plus CSF as the read-out stimuli [1,
5, 16]. This is an important observation for extrapolation of these
studies to clinical situations because it lays to rest some of the
concerns that combination biotherapy may accelerate exhaustion of
the stem cell compartment in patients receiving myeloablative therapy.
IV. Interleukin-1 and CSFs as Radioprotective Agents
Interleukin-1 has been shown to mediate a radioprotective action
when given to mice 20 h before what would otherwise be a lethal
dose of irradiation [12, 15]. This radioprotective effect cannot
be duplicated by preadministration of various CSF species. Using
BALB/c mice which are particularly susceptible to irradiation, we
observed that 0.2 ~g IL-1 given 20 h before 850 rads total body
irradiation completely prevented what would otherwise be 100% mortality
by 14 days, confirming earlier reports. However, we were able to
show that administration of IL-l or G-CSF after irradiation was
also signil1cantly radioprotective. While the mechanism of IL-l
radio protection remains controversial, our down observations suggest
that one mechanism may involve accelerated recruitment of primitive
Go stem cells into the cell cycle with expression of CSF receptors.
The administration of 750 rads total body irradiation to BALH/c
mice, which resulted in 80% mortality within 14 days, provided a
model in which we could test the interactions of IL-l and G-CSf
in reconstitution of hematopoiesis and in radioprotection. Recovery
of neutrophils to normallevels in irradiated mice was delayed for
up to 3 weeks; G-CSF administration begun immediately after irradiation,
and carried out for 3 weeks, accelerated recovery of neutrophils
and total hcmatopoietic cells, so that normal levels were observed
by 14 days [22]. IL-1 administration as a single dose prior to irradiation
resulted in accelerated recovery of hematopoiesis. When administered
daily postirradiation for 4 days, IL-1 reduced the nadir of blood
neutrophils and produced a degree of recovery or hematopoictic function
comparable to that seen with G-CSF administration (Fig. 3). A significant
synergistic interaction was noted when G-CSF was combined with IL-1
postirradiation, with up to a tenfold greater increase in blood
neutrophils, hematopoietic cells, and both stem and progenitor cells
populations in both spleen and bone marrow.
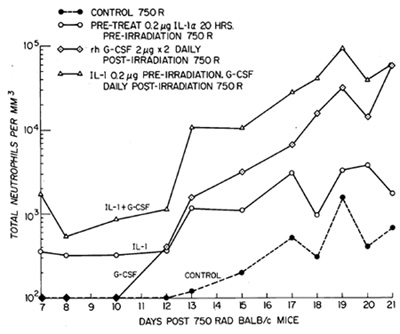
Fig.3. Absolute neutrophil counts in the peripheral blood
of Balb/c mice exposed to 750 rad total body irradiation and either
pretreated with rhI L-I alpha (0.2 µg/mouse i.p. 20 h before
irradiation) or treated with 2 µg rh-G-CSF beginning 2 h postirradiation
twice daily for 21 days, or the combination of IL-1 pre- and G-CSF
post-treatment. Five mice per group. Control mice (broken line received
saline only
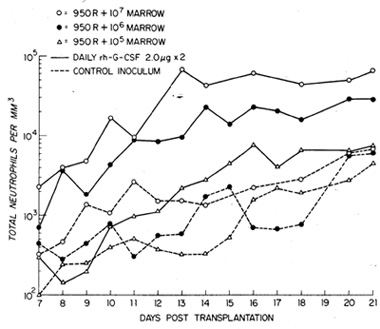
Fig.4. Absolute neutrophil counts in the peripheral
blood of B6D2F1 mice subject to 950 rads total irradiation and injected
with varying numbers of syngeneic bone marrow cells. with or without
subsequent twice daily administration of 2 µg rhG-CSF i. p.
Five mice were used per group
V. Interactions Between IL-1 and G-CSF in Autologous Bone Marrow
Transplantantion
Transplantation of autologous bone marrow into lethally irradiated
(950 rads) C3H/HeJ and B6D2F1 mice was undertaken using three different
dose levels of donor marrow cells, 100.000, 1million, and 10million
cells/mouse. The LD5o in such transplanted animals is observed at
dose levels of 1.0-2.5 x 1.million marrow cells/ mouse. It is generally
accepted that the delayed recovery in hematopoietic cel1s in the
bone marrow and spleen and in peripheral neutrophil counts was attributed
to limitations in the number of donor stem cel1s as measured by
the CFU-s assay. As can be seen in Fig. 4, the peripheral neutrophil
counts ref1ected a marrow cell dose-dependent difference in a return
to normal values. While this observation (which was also ref1ected
in bone marrow and spleen hematopoietic cell reconstitution) might
lead to the assumption that stem cell availability is the sole critcrion
for hematopoietic reconstitution, the administration of exogenous
G-CSF suggested additional variables in the transplant equation.
In all instances fol1owing autologous bone marrow transplantation
at the different dose levels, exogenous G-CSF administration enhanced
the recovery of neutrophils in the peripheral blood by three- to
tenfold. This in turn was accompanied by accelerated regeneration
of bone marrow and spleen hematopoietic progenitor ccl1s and of
pluripotential stem cel1s as measured by the CFU-s assay. Studies
in autologous bone marrow transplantation in cynomolgus monkeys
confirmed the ability of G-CSF to accelerate bone marrow regeneration
and recovery of peripheral blood neutrophil counts. These studies
indicated that higher doses of G-CSF were required to enhance the
recovery of hematopoietic parameters to normal, suggesting that
the role of G-CSF involved recruitment of earlier stem cel1s by
some indirect mechanism involving cytokine cascades or, alternatively,
reflecting the relative paucity of G-CSF receptors on primitive
hematopoietic cells. Based upon our in vitro studies indicating
that IL-1 upregulated the receptors for G-CSF and other CSF species
on primitive hematopoietic stern cells that were resistant to irradiation
and chemotherapy, we cultured bone marrow from mice treated with
5FU for 24 h with IL-1 prior to autologous transplantation in lethal1y
irradiated mice. Fol1owing transplantation, recipients received
1 L-1 alone for 4 days, G-CSF alone for 21 days, or IL-1 plus G-CSF,
and hematopoietic reconstitution was assessed. Preliminary studies
indicated that in vitro pretreatment of bone marrow enhanced bone
marrow reconstitution and accelerated recovery of peripheral neutrophil
counts in mice that subsequently received IL-1, G-CSF, or a combination
of IL-1 plus G-CSF in the post-transplant period. As measured by
reconstitution or total stern cel1s and progenitor cells, hematopoietic
recovery, and peripheral neutrophil counts, the most efficacious
combination involved pretreatment of bone marrow in vitro with IL-1,
followed by post-transplant treatment with a combination of IL-1
and G-CSF.
D. Conclusion
Numerous clinical trials are now underway using G-CSF and GM-CSF
in iatrogenic myelosuppressive situations associated with cancer
chemotherapy and irradiation therapy, and in conjunction with autologous
and al1ogeneic bone marrow transplantation. Efficacy is also under
study in congenital disorders of neutrophil production, and in myelodysplastic
syndromes and myeloid leukemias. The initial promise of thc CSFs
and interleukins suggests that they may provide a major new therapeutic
modality but only if we can develop a deeper insight into their
modes of action and interaction. The choice of the type or factor
to be administered will be influenced by the particulars of the
pathology being treat ed, making it meaningless to generalize as
to whether one factor is "better" than another. It is already clear
that the timing, dose, and route of administration of hematopoietic
growth factors arc important variables and efficacy will have to
be balanced against potential adverse side effects (which fortunately
have not emerged as serious limitations in the case of G-CSF and
GM-CSf trials). In addition, preclinical studies point to the value
of combination biotherapy using two or more factors administered
at the same time, or sequentially. Finally, there is a need to recognize
that proliferative processes arc self-limiting and physiological
mechanisms probably exist to counteract the action of hematopoietic
growth factors. Unraveling these issues wil1 undoubtedly occupy
the agenda of many future Wilsede Meetings.
References
1. Moore MAS ( 1988) The use or hematopoictic growth and differentation
factors for bone marrow stimulation. Ill: De Vita VT, Hellman S,
Rosenberg SA (cds) Important advances ill oncology 1988. Lippincott,
Philadelphia, pp 31-54
2. Welte K, Platzer E, Lu L, Gabrilovc JL, Levi E, Mertelsmann R,
Moore MAS (1985) Puriricatioll and biochemical characterization
or human pluripotent hematopoictic colony-stimulating factor. Proc
Natl Acad Sci USA 82.1526-1530
3. Souza L, Boolle TC, Gabrilove JL, Lai PH, Zsebo KM, Murdock DC,
Chazill VR, Bruszcwski J, Lu H, Chell KK, Barelldt J, Platzer E,
Moore MAS, Mertelsmann R, Welte K (1986) Recombinant human granulocyte
colony-stimulating factor; effects On normal and leukemic cells.
Science 232.61-65
4. Welte K, Bonilla MA, Ginio AP, Bone TC, Potter GK, Gabrilove
JL, Moore MAS, O'Reilly L Souza LM (1987) Rccombinant human granulocyte
colony stimulating factor. effects On hematopoiesis in normal and
cyclophosphamidetreated primates. J Exp Med 1645: 941-948
5. Moore MAS, Warrell W ( 1987) Inter Ieukill-1 and G-CSF synergism.
ill vivo slimulaliol1 of stem cell recovery al1d hematopoictic rcgcl1cratiol1
followil1g 5-fluorouracil treatmel1t il1 mice" Proc Natl Acad Sci
USA 84" 7134- 7138
6. Gabrilovc JL, Jakobowski A, Scher H, Slerl1bcrg C, Wong G, Grous
J, Yagoda A, Fain K, Moore MAS, Clarkson B, Oettgen HF, Alton K,
Welte K, Souza L (1988) Effect of granulocytc colony-stimulating
factor on neutropenia and associated morbidily due to chemotherapy
for transiliol1al-cell carcinoma of the urothelium" N Engl J Med
318:1414-1422
7. Jakubowski AA, Sou7a L, Kelly F, Fain K, Budman D, Clarkson B,
Bonilla MA, Moore MAS, Gabrilove JL (1989) Effects of human granulocyte
colony stimulating factor on a patient with idiopathic neutropenia
New Engl J Med 320 38 -42
8. Hernandez Bronchud MH, Scargge JH, Thatcher N, Crowther D, Souza
LM, AIton NK, Tesla NG, Dexter TM (1987) Phase 1/11 study of recombinant
human granulocyte colony-stimulating factor in patients receiving
intensive chemotherapy for small cell lung cancer" Br J Cancer 56:809
-813
9. Taylor K, Spitzcr G, Jagannath S, Dicke K, Vincent M, Souza L
(1988) rhG-CSF hastens granulocyte recovery in Hodgkin's disease
after high-dose chemotherapy and aulologous bone marrow transplant"
Exp Hcmaloll6"413 (abstr)
10. Lothrop Cl) Jr, Warren DJ, Souza LM, Jones JB, Moore MAS (1988)
Correction of canine cyclic hematopoiesis with recombinant human
granulocyte colony stimulating factor" Blood 72" 1324-1328
11.Bonilla MA, Gillio AP, Ruggiero M, Kernan NA, Brochstein JA,
f'umagalli L, Bordignon C, Vincent M, WeIte K, Souza LM, O'Reilly
RJ (1988) Corrcclion of neutropenia in patients with congenital
agranulocytosis with recombinant human granulocyte colony stimulating
factor in vivo" Exp Hemalol t 6 520 (abstr)
12. Oppenheim J, Kovacs E, Malsushima K, Durum SK (1986) There is
more than one interlcukin-1 Immunol Today 7:45-56
13. Lovhaug D, Pelus LM, Nordie EM, Boyum A, Moore MAS (1986) Monocyteconditioned
medium and inlcrleukin I in duce granulocylc macrophage colony stimulating
jaclor production in the adherent cell layer of murine bone marrow
cultures Exp Hematol 14:1037-1042
14. Bagby GC, Dinarello CA, Wallace P, Wagner C, Hefeneider S, McCall
E (1986) Interleukin I stimulates granulocyle macrophage colony
stimulating activity release by vascular endothelial cells J Clin
Invest 78:1316-1320
15. Neta R, Oppenhcim JJ (1988) Why should internists be interested
in il1lerleukin 1 ') Ann Intern Med 109.1-3
16. Moore MAS, Warren DJ, Souza L (1987) Synergistic interaction
between inlerIcukin-1 and CSF's in hemalopoiesis" In" Gale RP, Golde
l)W (eds) Recent advances in Ieukemia and lymphoma" UCLA symposium
on molecular and cellular biology Liss, New York, pp 445-458
17. Mochizuki l)Y, Eisenman J R, Conlon PJ, Larscn AD, Tushinski
RJ (1987) Interleukin I regulaled hemalopoielic activity, a role
previously ascribed to hemopoietin 1 Proc Natl Acad Sci USA 84:
5627
18. Warren DJ, Moore M AS (1988) Synergism among interleukin I,
interleukin 3, and inlcrleukil1 5 in lhc produclion of cosinophils
from primilivc hcmopoictic slem cclls J ImmunoI 140:94-99
19. Stanlcy ER, Bartocci A, Patinkin D, Rosendaal M, Bradley TR
(1986) Regulation of very primitive multipotcnt hemopoietic cells
by hemopoietin-1 Cell 45:667-674
20. Jubil1sky PI, Stal1lcy ER (1985) Purificalion of hcmopoietin-l"
a mullilineagc hcmopoictic growth factor" Proc Natl Acad Sci USA
82: 2764-2767
21. Gabrilove J, Welte K, Harris P, Platzer E, Lu L, Levi E, Mertelsmann
R, Moore MAS (1986) Pluripoielin alpha" a second human hcmatopoietic
colony-stimulating factor produced by the human bladder carcinoma
cell line 5637 Proc Natl Acad Sci USA 83: 2478-2482
22. Moore MAS, Welte K, Gabrilove JL, Souza LM ( 1986) In vivo action
of recombinant human G-CSF on chemotherapy or radiation myclosuppressed
mice" Blood 68 [Suppl l]:173a (abstr)
|





