|
* Supported by the Bundesminister für Forschung
und Technologie, FRG.
1 Universitäts-Kinderklinik Münster, FRG
2 Universitäts-KinderklinikHannover , FRG
3 Universitäts-Kinderklinik Düsseldorf, FRG.
A. Introduction
The number of children with acute myelogenous leukemia (AML) who
achieve remission and the number of long-term survivors have increased
in the last 10 years owing to intensified chemotherapy and better
supportive care. This report reviews nine pediatric studies, particularly
the German AML studies BFM-78 and BFM-83. A total of 294 children
with AML under 17 years of age entered the AML studies BFM-78 (n=151)
and BFM-83 (n = 143) between December 1978 and January 1986. The
second study is still open for patient entry. The treatment in the
first study consisted of a seven-drug regimen over a period of 8-10
weeks, together with prophylactic cranial irradiation, and was followed
by maintenance therapy with 6-thioguanine and cytosine arabinoside
(Ara-C) for 2 years and additional Adriamycin during the Ist year
[1]. In the BFM-83 study an 8-day induction with Ara-C, daunorubicin,
and YP16 precedes the BFM-78 protocol. The initial patient data
of the two studies are in general comparable age: median 9.11 and
9.3 years; sex: boys 54% and 52% ; WBC: median ( x 10ł/µl)
24.0 and 28.5; initial CNS involvement: 9% and 7%, respectively.
Extramedullary organ involvement (excluding liver and spleen enlargement)
was seen more often in the BFM-83 study (32%); it accounted for
only 18% of patients in the BFM-78 study. But the involvement of
bone, orbits, and kidney (7% in the BFM-83 study) was not evaluated
in the BFM- 78 study. The distribution of the F AB subtypes [2]
shows a higher proportion of the F AB M5 type (28%) in the BFM-83
study (only 21% in BFM- 78). In both studies the myeloblastic subtypes
Ml and M2 account for 20%-24% of patients, whereas the M3 and the
M6 subtypes were rarely seen (2%-4%). The overall results are presented
in Table 1. In the BFM- 78 study, 54 relapses (8 with CNS involvement)
occurred after a median follow-up time of 5.3 years (range 3.37.0
years). The life table estimations for an event-free survival (EFS,
total group) and an event-free interval (EFI, remission group) after
7.0 years are 38% (SO 4%) and 47% (SD 5%), respectively (Fig.1).
In the BFM-83 study, 25 relapses occurred (4 with CNS involvement)
after a median follow-up of 1.8 years (range 0.2-3.0 years). The
life table estimations are EFS 48% (SD 5% ) and EFI62% (SD 6%) (Fig.l).
Risk factor analysis shows that hyperleukocytosis (WBC > lOO x 10ł/µl)
is the main risk factor for early hemorrhage and/or leukostasis
(p < 0.001, X² test), for nonresponse (p<0.05, X˛ test), and
also for relapse (p= 0.08, log rank test). In addition, in the monocytic
subtypes M4 and M5, extramedullary organ involvement was a risk
factor for early hemorrhage and/or leukostasis (p<0.001) and also
for relapse (p=0.07, log rank test). The Ml subtype has the best
prognosis: EFS 55% (SD 7%) and EFI66% (SD 7%) after 7 years.
Table 1. Results of the AML studies BFM-78
and BFM-83, January 1986
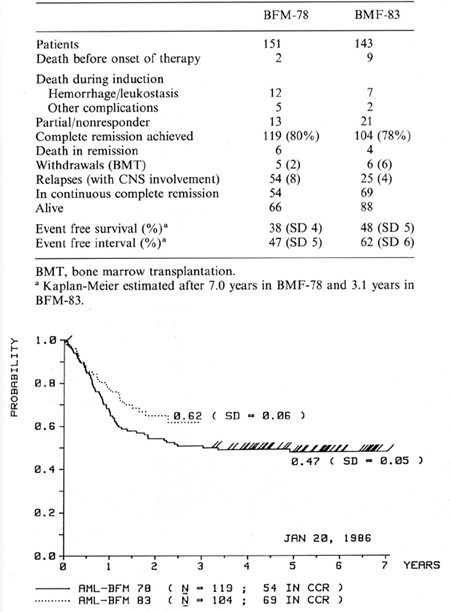
Fig.1. Probability of event-free interval in AML studies
BFM-78, and BFM-83. /, patients in CCR
(all patients of BFM- 78 study, last patient entered the BFM-83
study group).
CC R, continuous complete remission
B. Discussion
In most pediatric trials starting before 1976, the median duration
of complete remission was short less than 12 months; after 3 or
4 years, life table estimation for EFI was about 30% and for survival
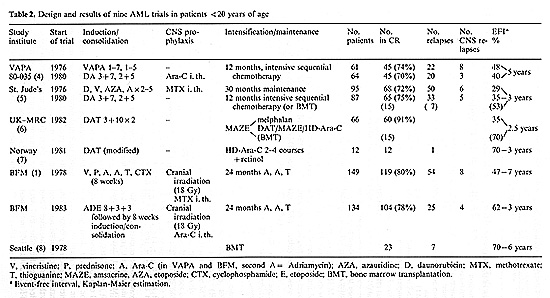
20% in the best studies [3]. Eight recent pediatric chemotherapy
protocols with high remission rates and good results are presented
in Table 2, together with one bone marrow transplantation (BMT)
trial. Even though the induction/consolidation regimens with two
to seven drugs differ considerably, they all include one of the
anthracyclines and Ara-C. Vincristine and prednisone were also administered
in the VAPA [4] and BFM [1] studies. The first St. Jude's study
[5] combined Ara-C with 6-azauridine. In consolidation of the BFM
studies, cyclophosphamide was given at least twice. In most studies,
remission was induced by relatively short and intensive therapy
with a seven-plus-three regimen (Ara-C plus daunorubicin), with
or without thioguanine, which induced a complete myelosuppression
and was followed by a therapy pause of approximately 3 weeks. In
contrast, the BFM78 study used a prolonged induction/consolidation
regimen for 8 weeks, which also caused severe bone marrow hypoplasia,
but in most cases the necessary therapy pauses were short. A new
strategy in intensive post-remission therapy called intensification
was initiated with the V APA-10 protocol [4] and is now part of
most of the new studies presented in Table 2. Lie et al. [7] reported
excellent results with high-dose Ara-C as postremission therapy
in a small group of children. The results of BMT, which is another
way of intensification in remission, are very encouraging, especially
in young patients [8]. In conclusion, new therapy strategies including
intensive induction regimens together with consolidation and intensification
or intensive maintenance with noncross-resistant drugs will improve
the treatment results in childhood AML and increase the proportion
of patients in long-term remission to 50%. The low incidence of
CNS relapses in the BFM studies indicates that prophylactic CNS
treatment early in remission can prevent CNS disease, and the increasing
number of long-term survivors emphasizes the need for effective
prevention of CNS relapse in pediatric patients. It still remains
to be seen whether prophylactic cranial irradiation together with
intrathecal methotrexate or Ara-C is necessary or whether systemic
treatment with Ara-C infusion or especially HD-Ara-C would produce
an effective liquor level. Although some results favor BMT, this
therapy is currently limited to patients with HLA-compatible donors,
and the long-term effects are unknown. Prospective comparisons ofBMT
with chemotherapy intensification or maintenance are necessary.
References
1. Creutzig U, Ritter J, Riehm H-J, et al. (1985) Improved treatment
results in childhood acute myelogenous leukemia. a report of the
German cooperative study AML-BFM 78. Blood 65.298-304
2. Bennett JM, Catovsky D, Daniel MT, et al. (1976) Proposals for
the classification of the acute leukaemias. Br J Haematol 33.451-458
3. Ritter J, Creutzig U, Riehm H, et al. (1984) Acute myelogenous
leukemia: current status of therapy in children. In. Thiel E, Thierfelder
S (eds) Recent results in cancer research, vol 93. Springer, Berlin
Heidelberg New York, pp 204-215
4. Weinstein H, Grier H, Gelber R, et al. (1987) Post remission
induction intensive sequential chemotherapy for children with AML
treatment results and prognostic factors. In. Biichner T, Schellong
G, Hiddemann Wet al. (eds) Haematology and blood transfusion, vo130.
Acute Jeukemias. Springer, Berlin Heidelberg New York, pp 88-92
5. Dahl GV, Kalwinsky DK, Mirro J, et al. (1987) A comparison of
cy to kinetically based versus intcnsive chemotherapy for childhood
acute myelogenous leukemia. In. Biichner T, Schellong G, Hiddemann
Wet al. (eds) Haematology and blood transfusion, vol 30. Acute leukemias.
Springer, Berlin Heidelberg New York, pp 83-87
6. Marcus RE, Catovsky D, Prentice HG, et al. (1987) Intensive induction
and consolidation chemotherapy for adults and children with acute
myeloid leukaemia Joint AML trial 1982-1985. In: Biichner T, Schcl1ong
G, Hiddemann Wet al. (eds) Haematology and blood transfusion, vol
30. Acute leukemia. Springcr, Berlin Heidelberg New York, pp 346-351
7. Lie SO, Slordahl SH (1987) High-dose cYtosine-arabinoside and
retinol in the treatment of acute myelogenous leukemia in childhood.
In. Biichncr T, Schel1ong G, Hiddemann Wet al. (eds) Haematology
and blood transfusion, vol 30. Acute leukemias. Springer, Berlin
Heidelberg New York, pp 399-402
8. Appelbaum FR, Thomas ED (1985) The role of marrow transplantation
in the treatmcnt of Icukemia. In: Bloomfield CD (ed) Chronic and
acute leukemias in adults. Nijhoff, Boston, pp 229-262
Additional participating members of the BFM-AML-Study Group
M. Neidhardt (Augsburg); G. Henze (Berlin); H.-J. Spaar (Bremen);
M. Jacobi (Celle); w. Andler (Datteln); J .-D. Beck (Erlangen);
B. Stollmann (Essen); B. Kornhuber (Frankfurt); A. Jobke (Freiburg);
G. Prindull (Göttingen); F. Lampert (Gießen); W. Brandeis (Heidelberg);
N. Graf (Hombilrg/Saar); H. Kabisch (Hamburg); G. Nessler (Karlsruhe);
H. Wehinger (Kassel); M. Rister (Kiel); F. Berthold (K61n-Univ.);
W. Sternschillte (K61n); 0. Sailer (Mannheim); C. Eschenbach (Marbilrg);
P. Giltjahr (Mainz); K.-D. Tympner (München-Harlaching); Ch. Bender-G6tze
(München-Univ.); St. Müller-Weihrich (M ünchen-Schwabing); R. J.
Haas (München v. Hailnersches Spital); A. Reiter (Nürnberg); W.
Ertelt (Stilttgart); D. Niethammer (Tübingen); G. Gaedicke (Ulm);
Th. Luthardt (Worms)
|

